Live Empowered.
TEA’s mission is to empower all those affected by EM to improve their quality of life.
Take TEA’s ‘A Patient’s Guide to Erythromelalgia‘ with you to your next doctor visit!!
The Patient Guide is a booklet covering all aspects of EM: symptoms, diagnosis, treatment, daily coping tips and more. It is a very important tool for anyone with EM (and their families and health care providers) to have on hand as they navigate improving their day-to-day lives.
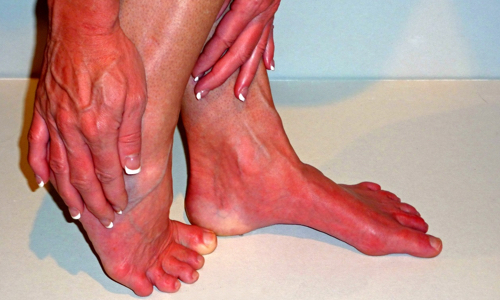
What is Erythromelalgia?
Erythromelalgia is a rare neurovascular condition that most commonly affects the feet, but may also occur in the hands, face, or other parts of the body. The term neurovascular means that both nerves and blood vessels are involved.

How do I find out if I have Erythromelalgia?
The three hallmark symptoms of EM are:
– Redness, caused by excessive blood flow to the area
– Heat, meaning that the skin will feel warm or hot to the touch, also due to increased blood flow
– Pain, which may range from mild tingling to severe burning
Additional symptoms may include swelling caused by the buildup of fluid in the affected body part, changes in perspiration, or purple discoloration when not actively flaring. EM flares are most commonly triggered by warmth, physical activity, or stress. A medical doctor may be able to give you an official diagnosis.

Stay up to date with our Social Media
If you or someone you know is affected with Erythromelalgia, you know how crucial it is to be updated on the latest medical information and research. Here at our organization, we strive to provide a safe and informative space online for patients and their families who are seeking guidance and support. Our Twitter, Facebook, and Instagram accounts are a valuable resource for those who want to stay informed on the latest news, medical research, and treatments. By connecting with us on social media, you can get the information you need to navigate this condition and advocate for the community. Don’t miss out on the opportunity to make a difference and stay connected with us!
Our Facebook page is almost always being updated!
Be sure to connect with our Instagram and YouTube pages below as well!
Join TEA
TEA currently has over 2,000 members residing in 17 countries. Stay up to date on the latest research findings by joining those affected by Erythromelalgia, their friends, family, and caretakers.
Frequently Asked Questions
Have more questions? TEA has compiled a list of answers to frequently asked questions to provide generalized and educational information.



